06/03/2022
Challenges in the Development of a Method for the Detection of Anti-PEGylated-aptamer Antibodies
Immunogenicity assays capable of properly determining the presence of anti-drug antibodies (ADA) and anti-PEG antibodies have been recognized of great importance due to the fact that pre-existing anti-PEG antibodies could have an impact on the efficacy of a drug coupled to polyethylene glycol (PEG).
AON-D21 is a clinical stage PEGylated L-configured oligonucleotide aptamer targeting complement factor C5a. In this study, the sponsor required a method to be developed for the determination of anti-AON-D21 and anti-PEG antibodies in human serum.
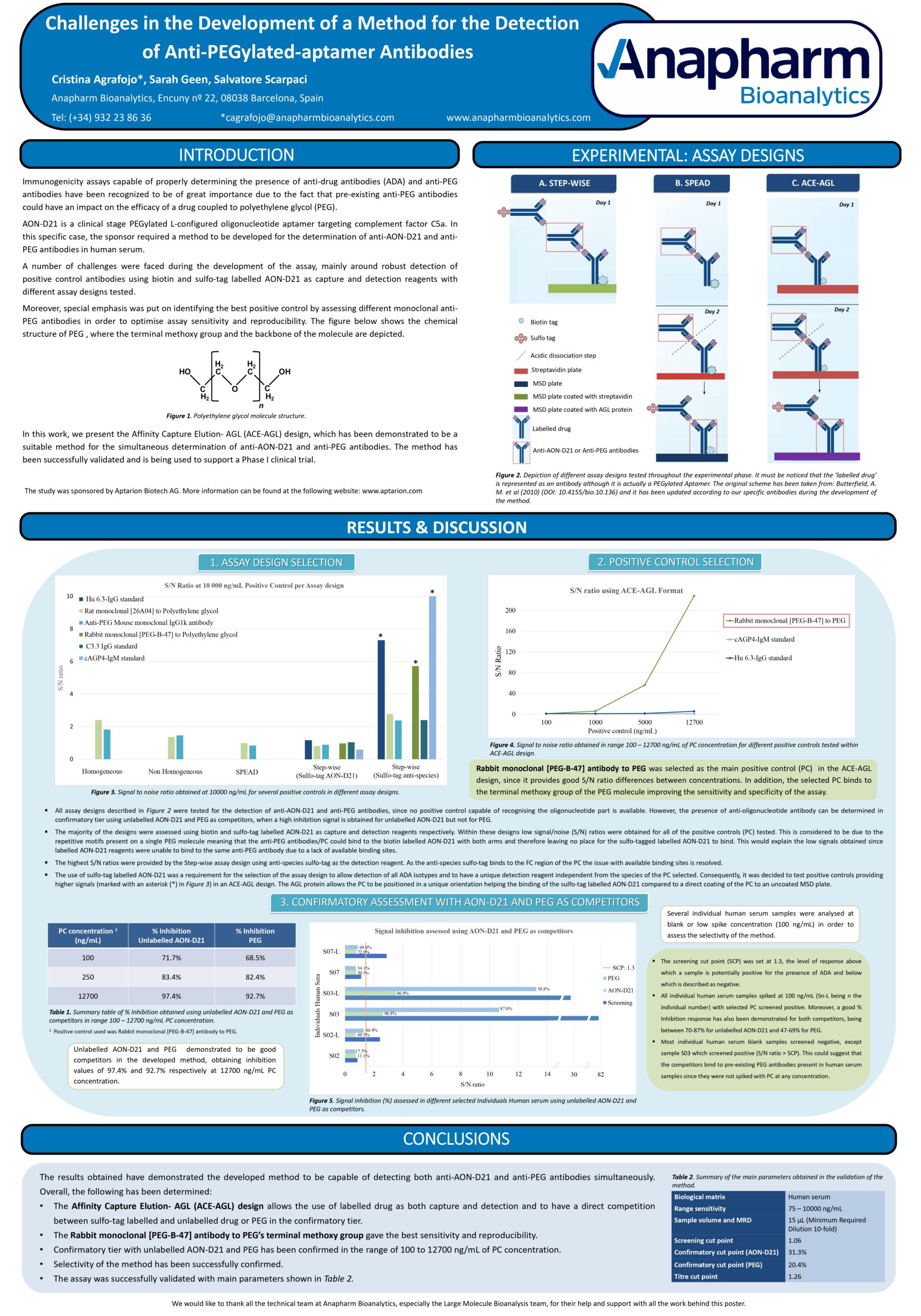
In the work we present to the 8th EBF Young Scientist Symposium, a number of challenges were faced during the development of the assay mainly around robust detection of positive control antibodies by using biotin and sulfo-tag labelled AON-D21 as capture and detection reagents. Several methods were evaluated with the most optimal assay design being the Affinity Capture Elution- AGL (ACE-AGL) format, which captures the ADA binding to immobilized biotin-AON-D21, followed by an acid elution step of ADA and finally the re-capture of these on a MSD plate coated with protein AGL. The ADA are then detected using sulfo-tag labelled AON-D21. In confirmatory assays, this is competed by unlabelled AON-D21 or PEG alone to identify the ADA binding site.
Special emphasis was put in identifying the best positive control by assessing different monoclonal anti-PEG antibodies in order to optimise assay sensitivity and reproducibility.
Finally, a suitable method capable of detecting both anti-PEG and anti-AON-D21 antibodies simultaneously was developed. The method has been validated and is being used to support a Phase I clinical trial in humans
You may find below the poster for your ready reference:

MORE NEWS
Webinar #1: Navigating Bioanalytical Challenges: from Small Molecules to Complex Peptides
In this 30-minute on-demand webinar, discover practical strategies to troubleshoot common analytical challenges, from small molecules to complex peptides, illustrated with real case studies from our laboratory.
Anti-PEG Antibodies: How Advanced Immunogenicity Testing Overcomes Detection Challenges
Detecting anti-PEG antibodies in clinical immunogenicity testing of PEGylated therapeutics presents unique bioanalytical challenges: from PEG’s repetitive structure causing low signal-to-noise ratios in standard assay formats, to selecting appropriate positive controls and distinguishing anti-drug from anti-PEG antibodies. This blog describes how the bioanalytical team at Anapharm Bioanalytics addressed these challenges during the development and validation of an immunogenicity assay using the Affinity Capture Elution–AGL (ACE-AGL) MSD immunoassay format.
Protein Digestion for LC-MS Bioanalysis: Comparing Standard and Kit-Based Methods
Quantitative bioanalysis of therapeutic proteins via liquid chromatography–mass spectrometry (LC-MS) typically relies on the analysis of surrogate peptides rather than intact proteins. Selecting the right digestion method impacts sensitivity, reproducibility, and cost. This blog compares three approaches tested in-house at Anapharm: standard trypsin digestion, the use of a kit with a thermostable immobilized trypsin, and a kit with ready-to-use reagents.

